How to Distinguish from Similar Species:Euplokamis dunlapae is similar shape but the body is more ovoid and flattened plus the tentacle branches (tentillae) are sparse and coil into bundles when contacted.
Geographical Range: Alaska to Acapulco, Mexico
Depth Range:
Habitat: Pelagic, especially in nearshore water
Biology/Natural History: Ctenophores have no nematocysts so they cannot sting. Instead, they have colloblast cells which produce a sticky substance to snare prey. Pleurobrachia bachei feeds on almost anything small enough for it to engulf, and may consume many copepods, eggs, and larval fish. It is a sit-and-wait predator. It swims horizontally in a semicircle while gradually extending its two tentacles until the tentillae hang down evenly spaced (photo). Then it stops and waits suspended with its mouth upright. When prey (usually a copepod) contacts the sticky net of tentillae and is snared, Pleurobrachia begins swimming forward while retracting its tentacles until the prey is close to its body. Then it begins rotating its body so the prey is brought around to the mouth and ingested. This species is the most common species of ctenophore likely to be encountered in the Northwest. Large aggregations may be seen in spring and summer. Individuals are hermaphrodites (both male and female). Eggs and sperm are released through the mouth and fertilized in the water. Not bioluminescent. May harbor a small red symbiotic amphipod, Hyperoche sp.
An extensive review article about cilia, swimming, neural circuitry, and behavior in ctenophores including Pleurobrachia can be found in Tamm (2014)
| Return to: | |||
| Main Page | Alphabetic Index | Systematic Index | Glossary |
References:
Dichotomous Keys:Flora and Fairbanks, 1966
Kozloff 1987, 1996
Smith and Carlton, 1975
General References:
Brusca
and Brusca, 1978
Harbo,
1999
Hinton,
1997
Kozloff,
1993
McConnaughey
and McConnaughey, 1985
Niesen,
1994
Niesen,
1997
Ricketts
et al., 1985
Sept,
1999
Smith
and Johnson, 1996
Wrobel
and Mills, 1998
Scientific Articles:
Greene, C.H., M.R. Landry, and B.C. Monger, 1986.
Foraging behavior
and prey selection by the ambush entangling predator Pleurobrachia
bachei.
Ecology 67: 1493-1501 Haddock, S.H.D. and J.F. Case,
1995. Not
all ctenophores are bioluminescent: Pleurobrachia.
Biol.
Bull. 189: 356-362
Haddock, Steven H.D., 2007. Comparative feeding
behavior of planktonic
cenophores. Integrative and Comparative Biology 47:6 847-853 Heimbacher
Goebel, Wyatt L., Sean P. Colin, John H. Costello, Brad J. Gemmell, and
Kelly R. Sutherland, 2020. Scaling of ctenes and consequences for
swimming performance in the ctenophore Pleurobrachia bachei. Invertebrate Biology 139:3 e12297. https://doi.org/10/1111/ivb.12297
Moss, A.G., 1991. The physiology of feeding in the
ctenophore
Pleurobrachia pileus. Hydrobiologia 216:
pp 19-25
Moss, A.G. and S.L. Tamm, 1981. Properties of the
unilateral ciliary
reversal response during prey capture by Pleurobrachia
(Ctenophora).
Biological Bulletin 161: p 308 (abstract)
Sleigh, M.A., 1968. Metachronal coordination of the
comb plates
in the Ctenophore Pleurobrachia. Journal
of Experimental Biology
28: pp 111-125
Tamm, Sidney L., 2014. Cilia and the life of
ctenophores.
Inverterate Biology 133:1 pp 1-46
Tamm, S.L. and A.G.Moss, 1985. Unilateral ciliary
reversal and
motor responses during prey capture by the Ctenophore Pleurobrachia.
J. Exp. Biol. 114: 443-461
Web sites:
General Notes and Observations: Locations, abundances, unusual behaviors:
This species is common pelagically during the summer.

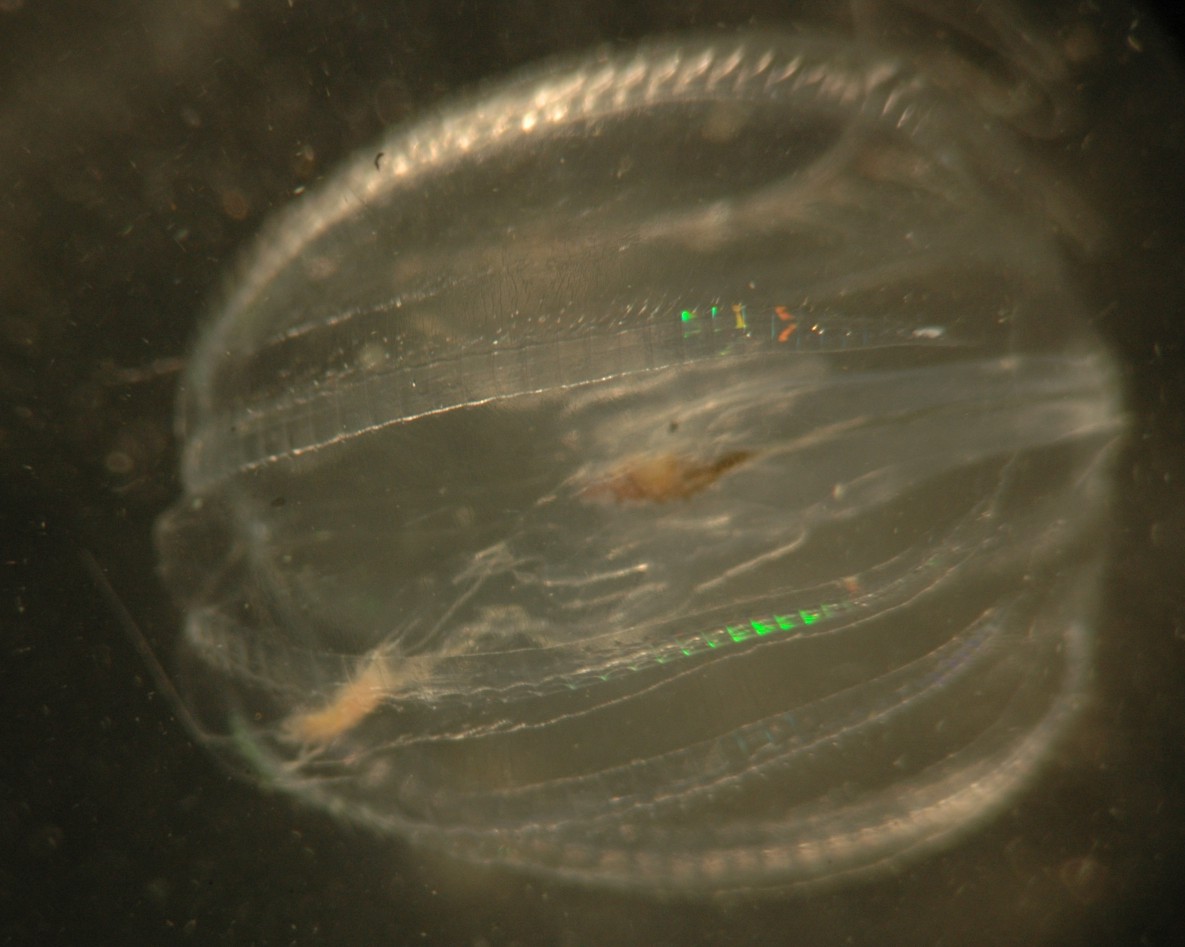
In this view the mouth is facing the camera and one tentacle
trails off to the left. The ends of the beating comb (ctene)
rows can be seen.

The long, retractable tentacles
have branches (tentillae)
along one side.

This closeup view of the aboral
end shows how the tentacle
sheaths angle out from the pharynx.
A partly retracted tentacle
can be seen on the lower left.
Authors and Editors of Page:
Dave Cowles (2007): Created original page
Jonathan Cowles (2007): Updated page with CSS